Breast Cancer Suite

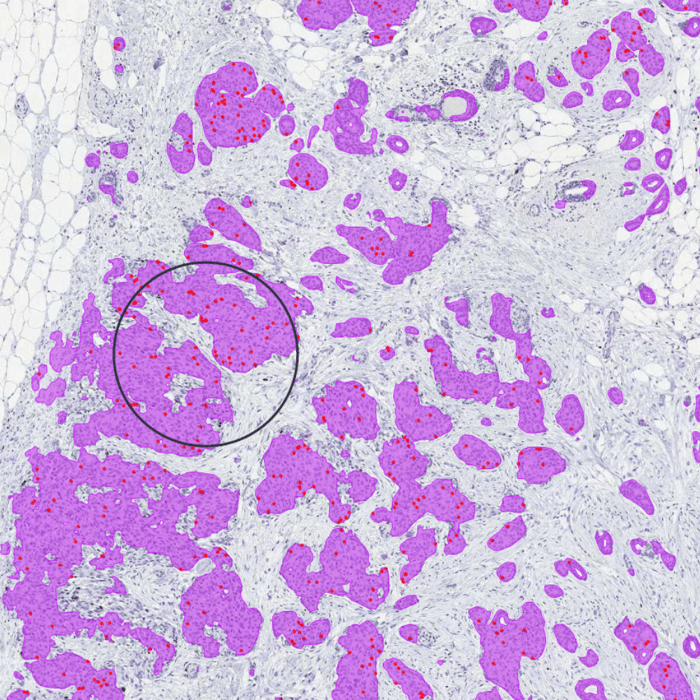
The AI model supports the histological grading of breast cancer in whole slide images (WSI) of H&E-stained tissue biopsies. It detects invasive carcinoma and ductal carcinoma in situ (DCIS) and scores the grading components mitotic count, tubulus formation, and nuclear pleomorphism. It has been designed based on the Nottingham Grading System.
Aiforia® Breast Cancer Grading is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.
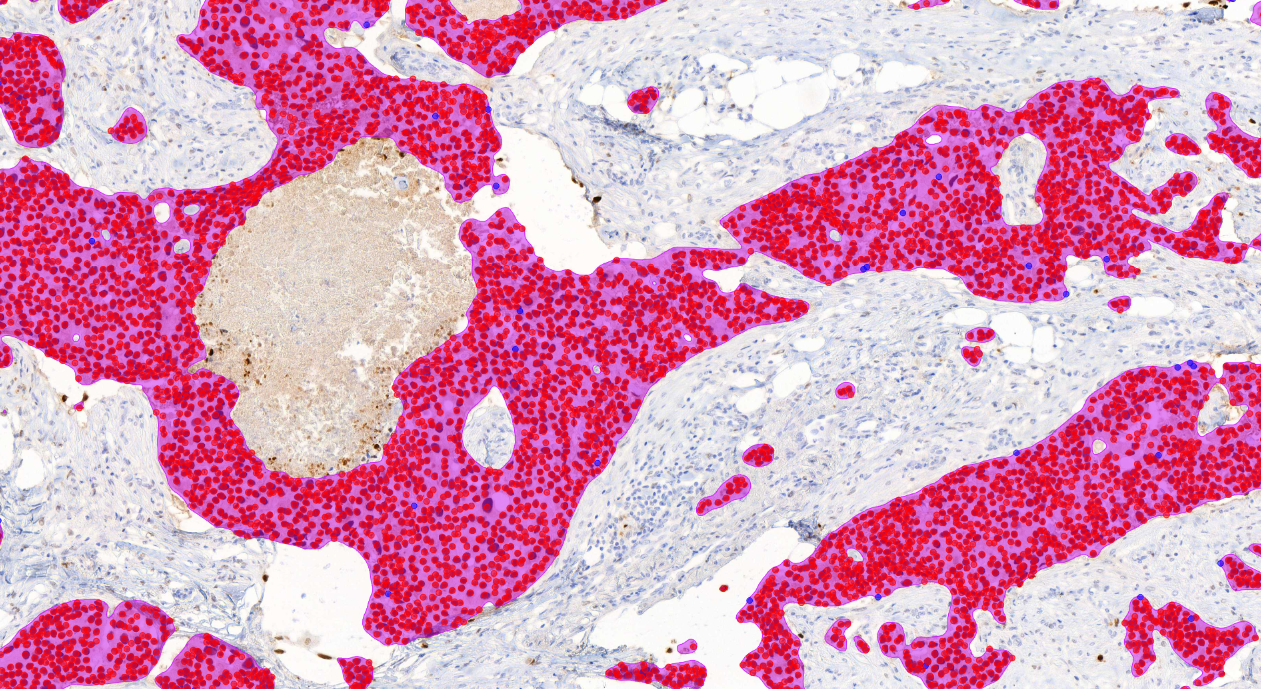
The AI model supports the detection of invasive carcinoma and scoring of ER positive and negative tumor cells from whole slide images (WSI) or from selected image areas of cancerous breast tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Saved time (up to 47% faster)
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
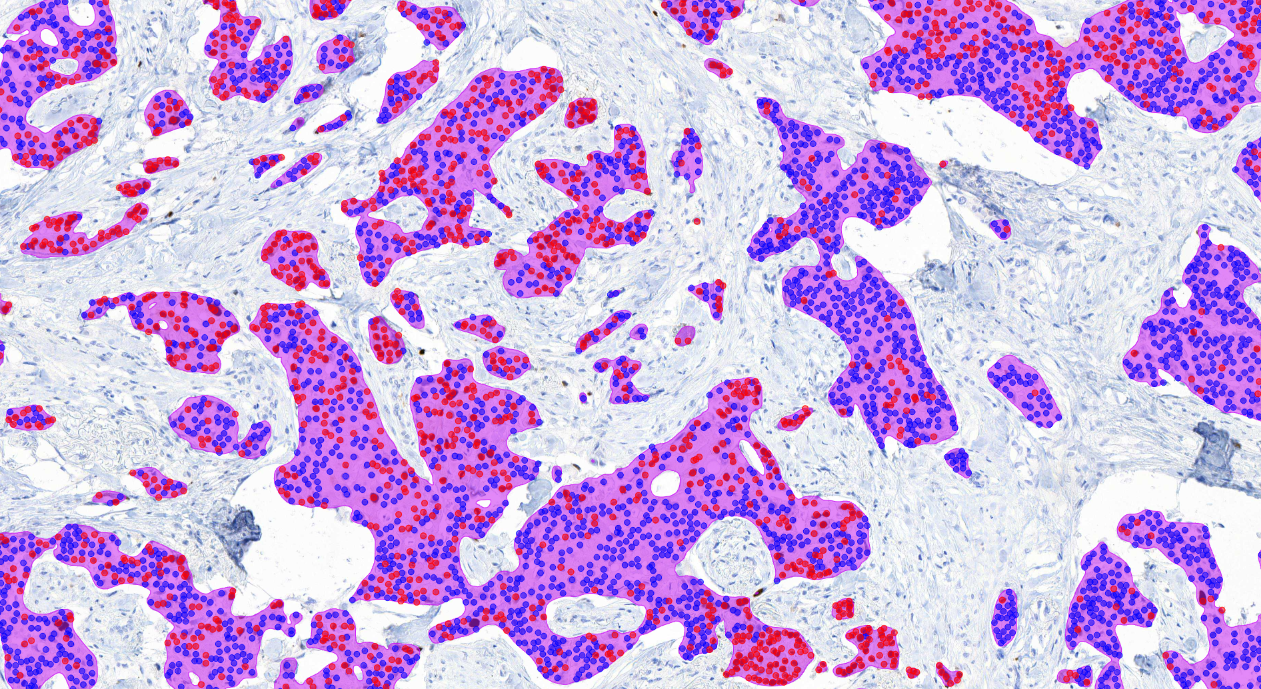
The AI model supports the detection of invasive carcinoma and scoring of PR positive and negative tumor cells from whole slide images (WSI) or from selected image areas of cancerous breast tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas

The Al model supports the detection of invasive carcinoma and scoring of HER2 positivity and intensity in tumor cells from whole slide images
(WSI) of breast tissue. The Al model has been designed according to CAP recommendations for HER2 analysis.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Improved diagnostic concordance by increasing pathologist agreement
CE-IVD marked for diagnostic use in EU and EEA countries (IVDR) and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
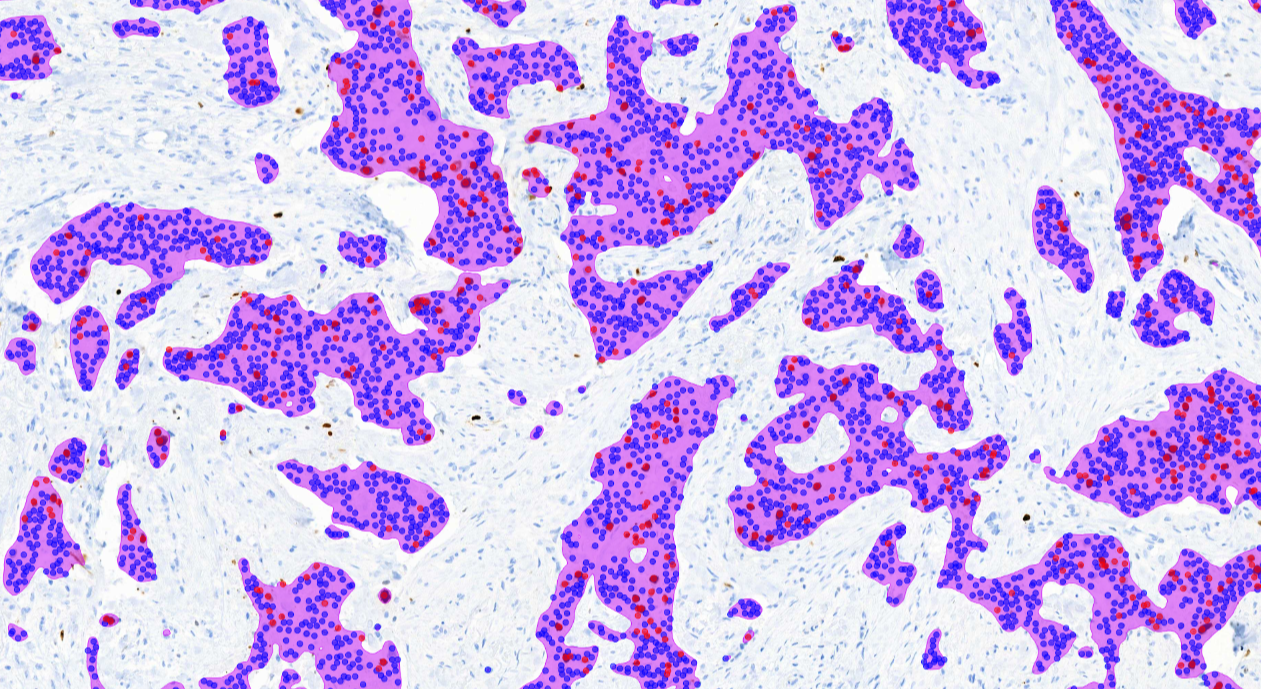
The AI model supports the detection of invasive carcinoma and scoring of Ki67 positive and negative tumor cells from whole slide images (WSI) or from selected image areas of cancerous breast tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Improved diagnostic concordance by increasing pathologist agreement
- Saved time in the whole slide image scoring (up to 54% faster)
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Breast Cancer Suite
Aiforia® Breast Cancer Grading

The AI model supports the histological grading of breast cancer in whole slide images (WSI) of H&E-stained tissue biopsies. It detects invasive carcinoma and ductal carcinoma in situ (DCIS) and scores the grading components mitotic count, tubulus formation, and nuclear pleomorphism. It has been designed based on the Nottingham Grading System.
Aiforia® Breast Cancer Grading is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.
Aiforia® Breast Cancer ER

The AI model supports the detection of invasive carcinoma and scoring of ER positive and negative tumor cells from whole slide images (WSI) or from selected image areas of cancerous breast tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Saved time (up to 47% faster)
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Aiforia® Breast Cancer PR

The AI model supports the detection of invasive carcinoma and scoring of PR positive and negative tumor cells from whole slide images (WSI) or from selected image areas of cancerous breast tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Aiforia® Breast Cancer HER2

The Al model supports the detection of invasive carcinoma and scoring of HER2 positivity and intensity in tumor cells from whole slide images
(WSI) of breast tissue. The Al model has been designed according to CAP recommendations for HER2 analysis.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Improved diagnostic concordance by increasing pathologist agreement
CE-IVD marked for diagnostic use in EU and EEA countries (IVDR) and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Aiforia® Breast Cancer Ki67

The AI model supports the detection of invasive carcinoma and scoring of Ki67 positive and negative tumor cells from whole slide images (WSI) or from selected image areas of cancerous breast tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Improved diagnostic concordance by increasing pathologist agreement
- Saved time in the whole slide image scoring (up to 54% faster)
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Aiforia® Lymph Node Metastasis

The AI model supports the detection and quantification of metastases of breast cancer, melanoma, and colorectal cancer in lymph nodes from whole slide images.
Aiforia® Lymph Node Metastasis is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.
AI for breast cancer diagnostics
Aiforia® Breast Cancer Suite offers a fully digital cockpit for breast cancer diagnostics, supporting the entire pathology workflow from sample analysis to diagnosis.
Lung Cancer Suite
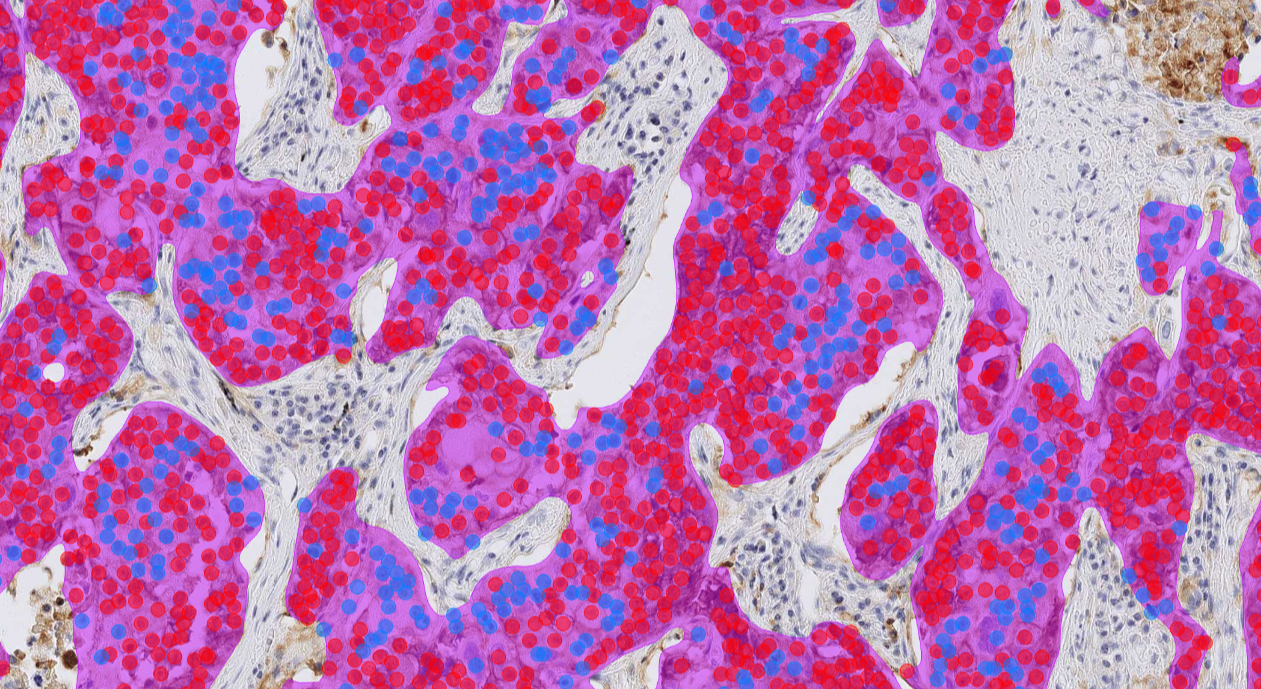
The AI model supports the detection of invasive carcinoma areas in patient samples and the scoring of PD-L1 negative and positive tumor cells in non-small cell lung (NSCLC) cancer cases.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Improved diagnostic concordance by increasing pathologist agreement
- Saved time (up to 46% faster)
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Lung Cancer Suite
Aiforia® Lung Cancer PD-L1

The AI model supports the detection of invasive carcinoma areas in patient samples and the scoring of PD-L1 negative and positive tumor cells in non-small cell lung (NSCLC) cancer cases.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Improved diagnostic concordance by increasing pathologist agreement
- Saved time (up to 46% faster)
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Prostate Cancer Suite
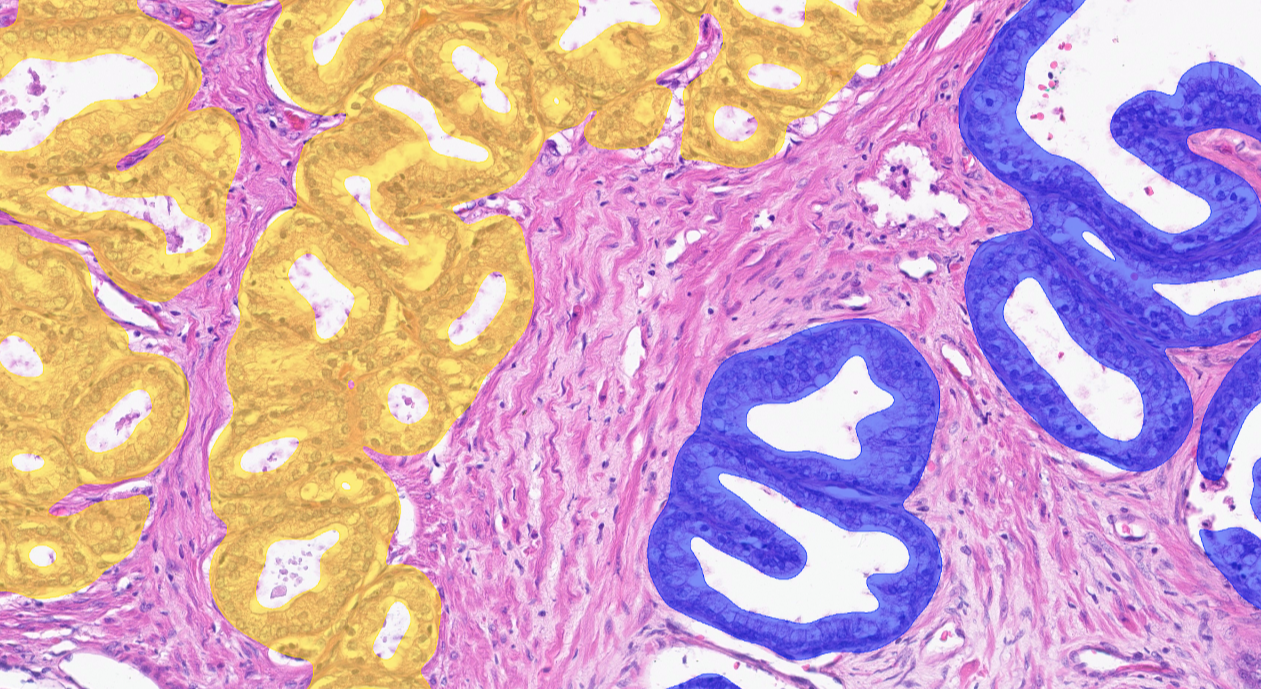
The AI model supports the detection of adenocarcinoma and assists Gleason grade grouping from whole slide images (WSI).
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Saved time (up to 36% faster)
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas

The AI model supports the detection of perineural invasion (PNI) in whole slide images (WSI) of prostate tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Saved time (up to 29% faster)
CE-IVD marked for diagnostic use in EU and EEA countries (IVDR) and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas

The AI model supports the detection of Gleason 4 cribriform pattern in whole slide images (WSI) of prostate tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Saved time (up to 54% faster)
CE-IVD marked for diagnostic use in EU and EEA countries (IVDR) and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Prostate Cancer Suite
Aiforia® Prostate Cancer Gleason Grade Groups

The AI model supports the detection of adenocarcinoma and assists Gleason grade grouping from whole slide images (WSI).
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Saved time (up to 36% faster)
CE-IVD marked for diagnostic use in EU and EEA countries, and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Aiforia® Prostate Cancer PNI

The AI model supports the detection of perineural invasion (PNI) in whole slide images (WSI) of prostate tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Saved time (up to 29% faster)
CE-IVD marked for diagnostic use in EU and EEA countries (IVDR) and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Aiforia® Prostate Cancer G4 Cribriform

The AI model supports the detection of Gleason 4 cribriform pattern in whole slide images (WSI) of prostate tissue.
Clinical Validation
- Showed excellent correlation with visual diagnosis
- Saved time (up to 54% faster)
CE-IVD marked for diagnostic use in EU and EEA countries (IVDR) and for Research Use Only (RUO) and Performance Studies Only (PSO) in all other market areas
Aiforia® Prostate Cancer HG-PIN

The AI model supports the detection of high-grade prostatic intraepithelial neoplasia (high-grade PIN) in whole slide images (WSI) of prostate tissue.
Aiforia® Prostate Cancer HG-PIN is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.
Colon Suite

The prognostic AI model identifies important histological features of colorectal cancer and provides a recurrence prediction estimate useful for treatment decisions. The AI model was developed and validated in collaboration with the Mayo Clinic.
Clinical Validation
- Verified and validated in the Aiforia® Platform using whole slide images from eight independent hospitals and patient cohorts
- Improved risk stratification as compared to the American Society of Clinical Oncology staging
Aiforia® Colorectal Cancer QuantCRC is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.

The AI model supports the detection and quantification of metastases of breast cancer, melanoma, and colorectal cancer in lymph nodes from whole slide images. The AI model was developed in collaboration with the University of Bern.
Aiforia® Lymph Node Metastasis is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.
Colon Suite
Aiforia® Colorectal Cancer QuantCRC

The prognostic AI model identifies important histological features of colorectal cancer and provides a recurrence prediction estimate useful for treatment decisions. The AI model was developed and validated in collaboration with the Mayo Clinic.
Clinical Validation
- Verified and validated in the Aiforia® Platform using whole slide images from eight independent hospitals and patient cohorts
- Improved risk stratification as compared to the American Society of Clinical Oncology staging
Aiforia® Colorectal Cancer QuantCRC is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.
Aiforia® Lymph Node Metastasis

The AI model supports the detection and quantification of metastases of breast cancer, melanoma, and colorectal cancer in lymph nodes from whole slide images. The AI model was developed in collaboration with the University of Bern.
Aiforia® Lymph Node Metastasis is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.
Gastric Suite
Gastric Suite
Aiforia® Gastric Cancer
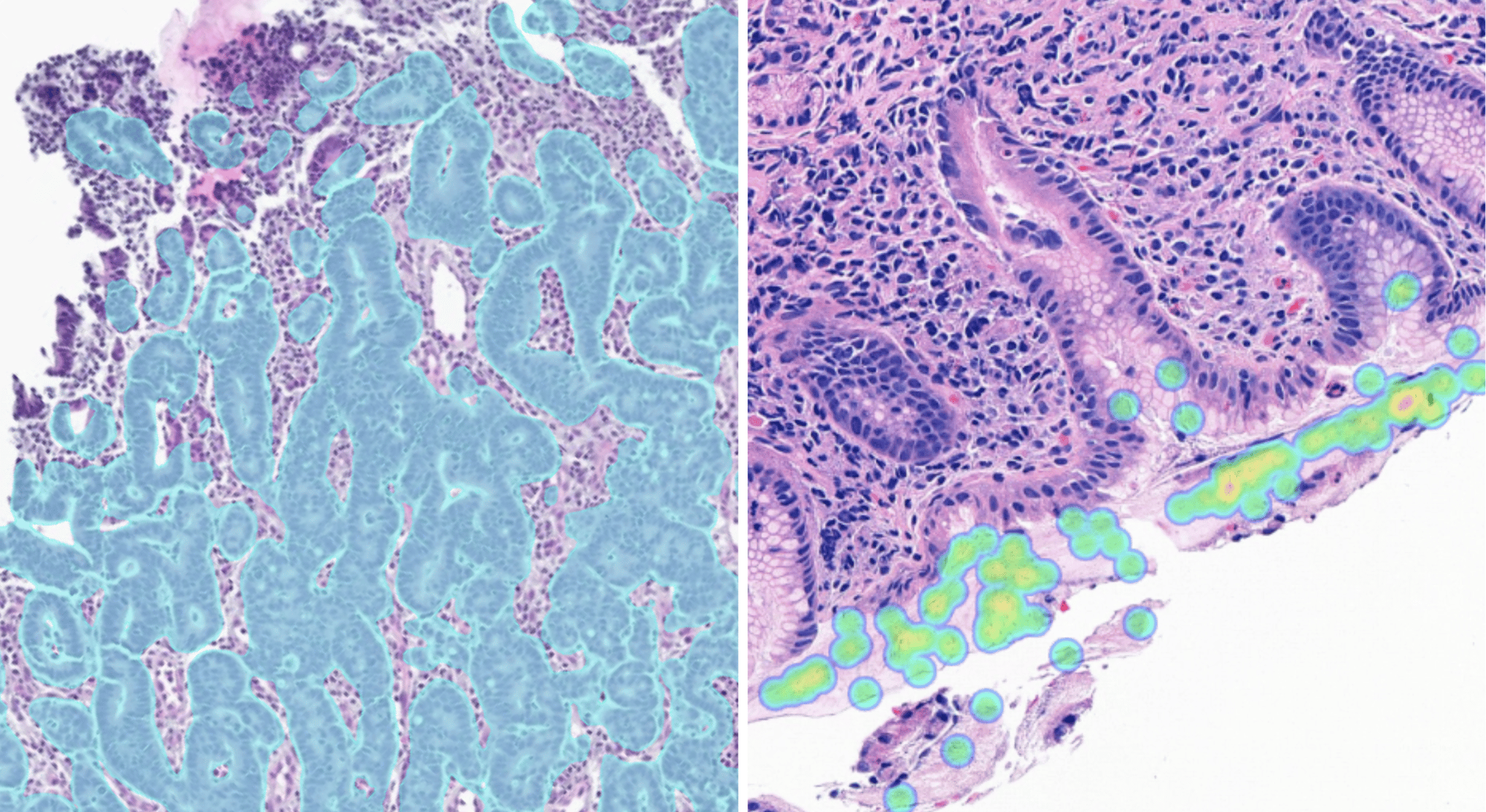
The AI model supports the detection of gastric cancer and Helicobacter pylori in whole slide images (WSI) of H&E-stained FFPE gastric tissue.
Aiforia® Gastric Cancer is currently for Research Use Only (RUO) and for Performance Studies Only (PSO) in all market areas.
Why use AI?
Increased efficiency
Diagnose more patients in less time by unbinding yourself from time-consuming, manual tasks.
Advanced precision
Improve diagnostic accuracy and be confident in your decision-making.
Improved consistency
Reduce variability to standardize your sample review and ensure democratized patient care.

What makes Aiforia unique?
Future-proof
Together with our customers, we continue to develop new groundbreaking AI models, with thousands of models already developed for research use. We’re committed to providing cutting-edge deep learning AI models designed to meet the evolving needs of our customers. As clinical diagnostics become increasingly complex, our advanced software will ensure you stay at the forefront of technology.
Transparency
Aiforia commits to keeping our AI algorithms transparent and understandable. You remain in control, see how analyses are conducted, and can even participate in the training process. AI models serve as first-read support while the expert verifies the results. We offer our customers fully quantifiable data and results that can easily be adjusted.
Ease of use
The Aiforia software requires no data scientists or coding expertise. Our team of experts integrates the solutions into existing IT infrastructures. We acknowledge that every lab is different, so we optimize each model for each lab’s individual needs and monitor their performance to deliver continuous improvement. Aiforia’s software allows customers to confidently scale up their operations while paying only for what they need.
Security
We take data privacy and security very seriously. We hold several globally relevant quality and security certifications, so our customers can be sure their sample and patient data is safe.* Customers determine what information is stored in the cloud and can opt for a private version.
*Our image analysis solutions are developed and maintained securely per ISO 27001 and SOC 2 Type II security standards, ISO 13485 quality standards, and IVDR requirements. In addition, we strictly adhere to GDPR requirements and all applicable HIPAA policies and procedures.
ON-DEMAND WEBINAR
Aiforia’s CE-IVD marked clinical AI models for Ki67 and PD-L1
Watch this recording for an intro and demo from pathologists Dr. Laury and Dr. Sjöblom on the Aiforia® Clinical AI Model for Lung Cancer; PD-L1 and Aiforia® Clinical AI Model for Breast Cancer; Ki67.
Why the cloud?
A cloud-based platform allows any laboratory with infinitely scalable digital pathology deployment.
Our services
Integration services
Integration can be straightforward. Aiforia solutions can be integrated with any existing laboratory infrastructure to enjoy the full benefits of a digitized workflow.
Cloud hosting services
Choose to host on our secure shared or private cloud environment. You can also opt to host on your own cloud provider (preferred partners include MS Azure and GCP).
Custom AI services
AI models developed by scientists, for scientists. With custom AI services, Aiforia's scientists build AI models for your specific needs.
AI for clinical diagnostics

BLOG POST
5 reasons to use AI in clinical diagnostics
What additional benefit does AI bring to the digitized pathology workflow in clinical diagnostics? Read our article to find out.
/Cancer%20JPEG/prostate_grading_after.jpeg?width=600&height=343&name=prostate_grading_after.jpeg)
INTERVIEW
Benefits of AI and digital pathology to the clinical lab
Clinical pathologists, Dr. Kevin Sandeman and Dr. Tuomas Mirtti, discuss the impact and benefits of AI and digital pathology in clinical diagnostics.

BLOG POST
How did the Mayo Clinic choose a vendor for AI in pathology?
What to consider when choosing a vendor for AI in pathology? Learn how the Mayo Clinic evaluated platform providers based on six key criteria.

BLOG POST
Digital pathology deployment in Veneto: 8 steps to success
What is required for pathology laboratories to go fully digital? Learn how the broadest public healthcare provider in the Veneto region did it.

NEWS
Aiforia obtains CE-IVD mark for AI-supported breast cancer diagnostics
Read our article to find out why the Aiforia Ki67 AI Model is a first-of-its kind diagnostic tool for pathologists to harness the full potential of their own expertise.
Request a demo
Discover the power of AI for image analysis
Find out how to enhance your image analysis work in diagnostic pathology, preclinical studies, or medical research. The demo will be tailored based on your interests.
The demo will help you understand:
- How AI-assisted image analysis can increase efficiency, precision, and consistency in the pathology workflow.
- The limitless possibilities Aiforia® Platform offers – both for research and clinical diagnostics – and the suitable use cases for your needs.
We can either demonstrate Aiforia’s image analysis solutions on your own images or any of our 400+ application examples (i.e., neuron quantification, automated tumor grading, NASH analysis, etc.).
Fill in the form, and one of our experts will contact you shortly to schedule the time.